Prefilled Syringes Market to Reach US$ 19.3 Billion by 2033, Expanding at 9.8% CAGR: Persistence Market Research
The prefilled syringes market is growing rapidly, driven by biologic drugs, chronic disease management, self-administration, and advanced injection technologie.
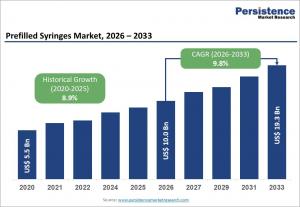
LONDON, UNITED KINGDOM, April 20, 2026 /EINPresswire.com/ -- The global prefilled syringes market is poised for substantial growth, with an estimated valuation of US$ 10.0 billion in 2026, expected to reach US$ 19.3 billion by 2033 at a compound annual growth rate (CAGR) of 9.8% during the forecast period. The market expansion is driven by rising demand for biologic drugs, increasing prevalence of chronic diseases, and the growing adoption of self-administered drug delivery systems. Prefilled syringes provide precise dosing and sterile delivery, making them a preferred choice for biologic therapies including monoclonal antibodies, hormones, and vaccines.
Pharmaceutical companies are increasingly incorporating prefilled syringes to enhance dosing accuracy, reduce medication preparation time, and minimize contamination risks. The expansion of global vaccination programs, coupled with the development of injectable treatments for conditions such as diabetes, rheumatoid arthritis, and autoimmune disorders, continues to boost market demand. Regulatory authorities and healthcare providers advocate for ready-to-use injectable formulations to enhance patient safety and treatment efficiency in both hospital and home care settings.
Download Your Free Sample & Explore Key Insights: https://www.persistencemarketresearch.com/samples/34583
Key Industry Highlights
Diabetes is anticipated to hold approximately 36% of market revenue in 2026, while oncology is expected to grow at the fastest CAGR of 11.5% through 2033, driven by increasing adoption of injectable biologic cancer therapies. North America is projected to dominate with a 39% market share in 2026, whereas Asia Pacific is expected to register the highest regional growth at 11.3% CAGR due to the expansion of vaccine manufacturing. Conventional prefilled syringes will continue to command around 64% revenue share in 2026, with safety prefilled syringes projected to grow the fastest, reflecting regulatory emphasis on needlestick injury prevention. Glass syringes are expected to lead in material preference with a 71% share, while polymer-based syringes are forecast to grow rapidly owing to durability and compatibility with advanced biologics.
Drivers of Market Growth
The rising prevalence of chronic diseases is a primary driver of market growth. According to the World Health Organization, chronic diseases account for nearly 74% of global deaths. Conditions such as diabetes, cardiovascular disorders, and autoimmune diseases require long-term injectable therapies. The International Diabetes Federation reported 537 million adults with diabetes in 2021, projected to reach 643 million by 2030, creating substantial demand for insulin delivery systems. Prefilled syringes simplify drug administration, improve dosing accuracy, and enhance patient convenience, particularly for insulin and biologic therapies. Home-based care trends and self-injection therapies further reinforce market growth.
The expansion of biologic drugs and global vaccination programs also fuels demand. Biologics represent over 40% of recent drug approvals, many requiring injectable delivery. Pharmaceutical companies increasingly integrate prefilled syringes into drug-device combination products to ensure consistent administration. UNICEF and Gavi report that more than 5 billion vaccine doses are administered annually worldwide, emphasizing the need for safe and efficient delivery systems. Recent FDA approvals of subcutaneous formulations for cancer therapies illustrate the shift toward injectable drug formats.
Challenges and Market Barriers
High manufacturing and material costs present a significant challenge. Prefilled syringes require precision filling, sterile packaging, and rigorous quality control. Glass syringes must adhere to stringent standards, and polymer alternatives demand compatibility testing. Rising raw material costs, particularly borosilicate glass and medical-grade polymers, add financial pressures. Regulatory complexity further complicates market entry, as prefilled syringes are classified as combination drug-device products requiring extensive testing for drug stability, sterility, and material compatibility.
Opportunities in Self-Administration and Emerging Markets
The trend toward home healthcare and self-injection therapies presents growth opportunities. Prefilled syringes allow patients to administer medications safely, ensuring accurate dosing and convenience for therapies such as insulin, monoclonal antibodies, and hormones. The global home healthcare market is projected to exceed US$ 600 billion by 2030, highlighting the potential for prefilled syringes in patient-centered care. Emerging markets in Asia-Pacific, Latin America, and the Middle East are expanding healthcare infrastructure and pharmaceutical manufacturing, driving demand for high-volume syringe solutions. Technological innovations, including safety syringes and advanced polymer designs, enhance market prospects by improving patient and healthcare worker safety.
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/34583
Regional Insights
North America is expected to maintain dominance due to advanced pharmaceutical manufacturing and regulatory frameworks supporting self-administration. Europe ranks second, benefiting from harmonized regulations, biotechnology research, and vaccination infrastructure. Asia Pacific is projected to grow fastest, driven by healthcare infrastructure expansion, vaccine production, and government initiatives supporting prefilled syringe adoption.
Market Segmentation
By Product Type
Conventional Prefilled Syringes
Safety Prefilled Syringes
By Material
Glass
Polymer
By Application
Diabetes
Rheumatoid Arthritis
Oncology
Vaccination
Cardiovascular Diseases (CVDs)
By Region
North America
Europe
East Asia
South Asia & Oceania
Latin America
Middle East & Africa
Get Custom Insights Designed for Your Businecss: https://www.persistencemarketresearch.com/request-customization/34583
Competitive Landscape
The global market is moderately consolidated, with key players including Becton Dickinson, Gerresheimer, Terumo, West Pharmaceutical Services, and SCHOTT AG, leveraging regulatory expertise, integrated production, and strong pharmaceutical partnerships. Regional players like Nipro Corporation, Ypsomed, and NABI target specialized segments and emerging markets. Strategic investments, partnerships, and technological innovations, including safety and polymer syringe advancements, continue to drive competition and market growth.
Conclusion
The prefilled syringes market is experiencing robust growth, driven by increasing biologic drug demand, chronic disease prevalence, and the shift toward self-administration and home healthcare. While challenges such as high production costs and regulatory complexity exist, innovations in materials, safety mechanisms, and regional expansion opportunities position the market for sustained growth through 2033.
Read Related Report:
Vaginosis Rapid Testing Market: The global vaginosis rapid testing market grows from US$ 0.4 Bn in 2026 to US$ 0.6 Bn by 2033, at an 8% CAGR during forecast 2026–2033.
Peripheral Vascular Devices Market: The global peripheral vascular devices market is projected to grow from US$12.6 Bn in 2026 to US$17.5 Bn by 2033, at a CAGR of 4.8%.
Persistence Market Research
Persistence Market Research Pvt Ltd
+1 646-878-6329
email us here
Visit us on social media:
LinkedIn
Instagram
Facebook
YouTube
X
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.